
CytoGUARD® LA Lauric Arginate Antimicrobial
GRAS Certified Antimicrobial Made from Naturally-derived Ingredients
CytoGUARD® LA is a powerful, food-grade antimicrobial agent with Lauric Arginate (LAE) as the key active ingredient. Optimized for maximum stability, it is a broad-spectrum antimicrobial effective against foodborne pathogens such as Listeria, E. Coli, Salmonella, and spoilage bacteria.
Highlights
- Lauric Arginate (LAE) is GRAS certified and approved for use in the U.S., Canada, and Mexico, among other countries
- LAE is a powerful antimicrobial known to reduce various foodborne pathogens and spoilage bacteria
- Provides the initial kill step in the food production process and packaging equivalent to in situ pasteurization, significantly reducing bacteria loads
- When combined with other natural preservatives, it can substantially improve food safety and shelf life
- Does not affect the taste or the sensorial properties of the food
- CytoGUARD® LA is a highly cost-effective antimicrobial agent
- Safe to use in food applications



Features
- Kills bacteria on contact (best used as a surface treatment)
- Can be used with other inhibitors to extend shelf life
- Easy to use
- Low cost in use
- Clear flavorless liquid
- Stable up to one year
- Made in the U.S.
Benefits
- Broad-spectrum antimicrobial
- Effective tool for pathogenic bacteria control providing a high degree of food safety
- Effective against spoilage bacteria which is an important step to ensure food protection and food quality
- Activity over a wide pH range (3 to 7)
- High partition coefficient (>10) means the agent concentrates in the water phase of products, where most bacterial activity occurs
- Does not affect the appearance or flavor of the food
- Low usage levels
- Processing aid status in some applications
- Efficacy supported by extensive internal studies and published documents (including USDA) and can be validated with customer products at A&B Ingredients micro lab
Applications
- Raw meat and poultry
- Ready-to-eat fresh cuts of meat and poultry
- Frozen seafood
- Dairy
- Fresh produce
Regulations
LAE is GRAS Certified:
- Granted by a panel of FDA experts in the U.S. through scientific procedures set forth under 21 CFR §170.30(b)
- Approved in Canada, EU, Mexico, Australia/NZ
What Is Lauric Arginate?
Ethyl lauroyl arginate is a food preservative, which has been known by many names. In the U.S. the common name is Lauric Arginate. Ethyl lauroyl arginate is also known as Lauramide arginine ethyl ester, Ethyl Nα-lauroyl-L-arginate, and Lauric Arginate ethyl ester. Ethyl lauroyl arginate is a potent antimicrobial compound derived from natural sources, specifically Lauric Acid, Arginine, and Ethanol. Ethyl lauroyl arginate is a unique ingredient made from the reaction of Lauric acid (from palm oil or coconuts) with arginine (an amino acid) in the presence of ethanol.
CytoGUARD® LA is a range of products based on Ethyl lauroyl arginate (Lauric Arginate) optimized for use in food and beverage products. CytoGUARD® LA was the pioneering product in a line of food preservatives developed and manufactured by A&B Ingredients under the brand name CytoGUARD®.
Broad-Spectrum Antimicrobial Properties
Ethyl lauroyl arginate is a broad-spectrum antimicrobial and has a remarkable ability to combat a wide range of microorganisms. Ethyl lauroyl arginate is effective against both . Ethyl lauroyl arginate is a potent bactericidal agent against foodborne pathogens such as E. coli, Salmonella, Listeria, and Norovirus. Ethyl lauroyl arginate is also effective against spoilage bacteria, like Carnobacterium and Lactobacillus. Ethyl lauroyl arginate can act as a "kill step," which ultimately reduces the log count of bacteria. In addition, Lauric Arginate greatly enhances the appearance of food products during refrigerated storage. This is due to Lauric Arginate's activity against lactic acid bacteria, which are known as primary agents in the deterioration of food products.
Efficacy Against Yeast and Molds
Ethyl lauroyl arginate is also proven to kill yeast and molds. This "kill step" concept can be compared to the "cold pasteurization." Notably, using Lauric Arginate antimicrobial for food safety has had a profound impact on the meat, fish, and seafood industries by enhancing the manufacturer's ability to ensure product safety and extend shelf life.
Ingredient Safety
Ethyl lauroyl arginate is a safe ingredient. Ethyl lauroyl arginate is hydrolyzed in the human body by chemical and metabolic pathways, which quickly breaks the molecule into natural components.
How Does Lauric Arginate Work?
Lauric Arginate, also known as Ethyl lauroyl arginate, is an antimicrobial with a broad-spectrum activity primarily used as a preservative in various food products. It inhibits the growth of microorganisms, ultimately extending the shelf life and ensuring the safety of food products.
Lauric Arginate is an amino acid-based cationic surfactant with low toxicity and antimicrobial activity. Lauric Arginate is widely used as a food preservative and a component of food packaging.
What Is Cationic Surfactant?
Cationic surfactants are characterized by a hydrophilic (water-attracting) head with a positive charge and a hydrophobic (water-repelling) tail. The positive charge on the head of the surfactant makes it attracted to negatively charged surfaces, such as cell membranes and proteins. When Lauric Arginate encounters a cell wall, it can destabilize the membrane, causing it to lose its integrity. Lauric Arginate interferes with membrane transport mechanisms across the cell wall and disrupts cellular metabolism, and the cell reproductive cycle. The "kill," otherwise known as the bactericidal effect, takes place almost instantaneously.
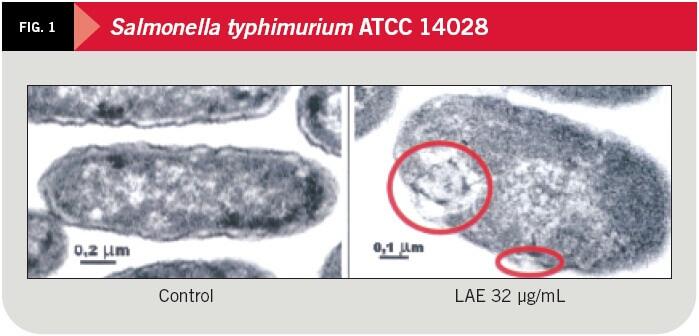
Lauric Arginate Mode of Action
- Disrupts lipid bilayer of plasma membrane, depicted in Figure 1 below.
- Disruption of membrane:
- Affects Cell metabolism
- Affects Cell cycle
- Causes leakage of intracellular components
- Interacts with bacterial DNA, resulting in structural changes
Extending Shelf Life Through Synergistic Action
Later research has shown that a small percentage of the cells remain weak and damaged and are more susceptible to physical (heat or freezing) or other antimicrobial interventions used. Manufacturers can achieve even better shelf life for food products when they combine Lauric Arginate with a bacteriostatic preservative. A&B Ingredients has developed CytoGUARD® STAT-N, a natural preservative that offers synergistic effect when used in combination with Lauric Arginate antimicrobial to further improve product's safety and extend shelf life.
Common antimicrobials used in the meat industry that have potential synergistic action with Lauric Arginate include lactate and diacetate, vinegar, and citrus extracts, all USDA-approved antimicrobials.
Lauric Arginate is generally considered to have minimal impact on the flavor and texture of food products. It can be used at low concentrations to achieve the desired preservative effect without altering the sensory attributes of meat, poultry, fish, or seafood products.
Regulations
Lauric Arginate, also known as Ethyl lauroyl arginate, is a food-grade antimicrobial agent approved for use as a food preservative. Lauric Arginate is considered safe for consumption when used within regulatory limits established by food safety authorities, such as the U.S. Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA). Food manufacturers use Lauric Arginate as a valuable tool to improve the safety and shelf stability of a wide range of food products, including meat, poultry, seafood, and more.
Lauric Arginate was affirmed as GRAS (generally recognized as safe) by the FDA in 2005. In 2007, Lauric Arginate was approved as a food preservative by the EFSA. Lauric Arginate is ideal for use as a food preservative in RTE (ready-to-eat) meat and poultry products. The approved dosage is up to 200 ppm.

MIC Data
Minimum Inhibitory Concentrations Data For Lauric Arginate
Minimum inhibitory concentrations (MICs) are defined as the lowest concentration of an antimicrobial that will inhibit the growth of a given strain of bacteria.
When choosing an antimicrobial, there are many factors to be considered, including cost, safety, convenience, and effectiveness. MIC data is useful in determining the effectiveness of an agent in inhibiting microorganism growth. Lauric Arginate is an excellent antimicrobial, demonstrated by Figure 1, as it is highly effective at low levels. In order for other antimicrobials to achieve the same desired effect, they would have to be used in much higher concentrations which has an array of detrimental effects.

Learn more about , studies, and reports.
SLIC® Application Method
The preferred method of application of CytoGUARD® LA is through the SLIC® (Sprayed Lethality in Container).
What Is SLIC®?
SLIC® is a patented process designed to improve and ensure the safety of RTE (ready-to-eat) processed meat products against contamination by pathogenic bacteria like Listeria monocytogenes. The SLIC® application method utilizes the unique approach of treating the surface of the meat products by adding an antimicrobial solution to the purge at the time of packaging. Due to this unique approach, the amount of antimicrobial agent used is based on the surface area rather than the weight of the product, ensuring that no antimicrobial agents are wasted. Unlike other antimicrobials, like potassium lactate, which is used internally in products as a part of the product's formulation, in the SLIC® application, the amount of antimicrobial used is sparse, thus having minimal effect on flavor while ensuring food safety.
Enhanced Antimicrobial Performance
Unlike spays and dips used to apply antimicrobial agents to meat products, with SLIC® application, the antimicrobial has almost unlimited time to perform its functions. Due to the SLIC's unique antimicrobial application inside the vacuum packaging, there is no antimicrobial waste from drip, spillage, evaporation, or dilution.
What Is Purge?

Widespread outbreaks of Listeria infections in the 1990s were primarily linked to deli meats and hot dogs. The Center for Disease Control (CDC) conducted research and analysis to determine the source of Listeria contamination in hot dogs, and they identified the purge in hot dog packaging as a likely source of the pathogens.
Purge is created over time, as a combination of water and meat proteins drain from the product and naturally accumulates on the exterior and within the spaces of vacuum packaging systems.
After these initial findings, the United States Department of Agriculture (USDA) Agricultural Research Service (ARS) conducted further research and discovered that using the purge to test for Listeria in hot dog packaging was more than five times more likely to yield a positive test result compared to testing the meat itself. These findings emphasize the significance of purge as a potential source of Listeria contamination in hot dogs and the importance of addressing it to ensure food safety.
Lauric Arginate Demonstrates Optimal Results
The ARS tested different antimicrobials at various strengths and volumes to determine the optimal solution to ensure the food safety of vacuum-sealed meat products. The best results were achieved using Lauric Arginate. Lauric Arginate is now available for use as a food preservative under the brand name CytoGUARD® LA, distributed by A&B Ingredients.
Learn more about , studies, and reports.

Publications
The efficacy and safety of Lauric Arginate is well researched. You can discover numerous Lauric Arginate publications on this page, along with a comprehensive list of efficacy tests conducted by globally renowned institutions for Lauric Arginate.
Synonyms:
- Lauric Arginate
- Lauric Arginate Ester
- Na-lauroyl Arginate ethyl ester
- L-arginine, Na-lauroyl ethyl ester
- LAE
- Ethyl Lauroyl Arginate HCI
Source: Science Direct – International Journal of Food Microbiology, Volume 315, 16 February 2020, by Qiumin Ma, P. Michael Davidson, Qixin Zhong.
Evaluation Of A Novel Antimicrobial (Lauric Arginate Ester) Substance Against Biofilm Of Escherichia Coli O157:H7, Listeria Monocytogenes, And Salmonella sppSource: Wiley – Institute of Food Science and Technology, Volume 52, Issue 9, 26 June 2017, by Mohammad Sadekuzzaman, Sungdae Yang, Hyung-Suk Kim, Md. Furkanur Rahaman Mizan & Sang-Do Ha
The Lowdown on Lauric ArginateSource: Food Quality & Safety, 25 May 2017, by Gil Bakal, Managing Director, A&B Ingredients and Alex Diaz, Export Manager, Vedeqsa
MSU Researchers Fight Poultry PathogensSource: Mississippi Agricultural and Forestry Experiment Station, 8 January 2016 | by Sarah Buckleitner
Investigating The Control Of Listeria Monocytogenes On Alternatively-Cured Frankfurters Using Natural Antimicrobial Ingredients Or Post-Lethality InterventionsSource: Meat Science, Volume 97, Issue 4, August 2014, by Nicolas A. Lavieri, Joseph G. Sebranek, Byron F. Brehm-Stecher, Joseph C. Cordray, James S. Dickson, Ashley M. Horsch, Stephanie Jung, Elaine M. Larson, David K. Manu, Aubrey F. Mendonca
Antimicrobial Properties Of Lauric Arginate Alone Or In Combination With Essential Oils In Tryptic Soy Broth And 2% Reduced Fat MilkSource: International Journal of Food Microbiology, Volume 166, Issue 1, 16 August 2013, Pages 77-84, Qiumin Ma, P. Michael Davidson, Qixin Zhong
Control Of Listeria Monocytogenes On Commercially-Produced Frankfurters Prepared With And Without Potassium Lactate And Sodium Diacetate And Surface Treated With Lauric Arginate Using the Sprayed Lethality in Container (SLIC®) Delivery MethodSource: Meat Science, June 2010, by A.C.S Porto-Fett, S. Campano, J. L. Smith, A. Oser, B. Shoyer, J.E. Call, J. Luchansky
Nalpha-lauroyl-L-arginine ethylester Monohydrochloride Reduces Bacterial Growth In Pasteurized MilkSource: Journal of Dairy Science, September 2009, by N. H. Woodcock, B. H. Hammond, R.D. Ralyea, K. J. Boor
Control Of Listeria Monocytogenes By Lauric Arginate On Frankfurters Formulated With Or Without Lactate/DiacetateSource: Journal of Food Science, 2009 Aug;74(6):M237-41, by E.M. Martin, C. L. Griffis, R.Y. Lary Jr., K. Vaughan, C. A. O'Bryan, E. Friedly, J. Marcy, S.C Ricke, P. Crandall
Inactivation of Listeria monocytogenes on Hams Shortly after Vacuum Packaging by Spray Application of Lauric ArginateSource: Journal of Food Protection, Volume 72 (12), Pages 2517-2523, 2009, by P. J. Taormina, W.J. Dorsa
Viability Of Listeria Monocytogenes On Commercially-Prepared Roast Beef Logs, Turkey Breast Logs, And Frankfurters Surface Treated With Lauric Arginate And Stored At 4°C For 24 HoursSource: Abstracts of the Annual Meeting of the International Association for Food Protection (P2-24), p148, 2007, by Luchansky, J. B., J. E. Call, J. L. Smith, J. L. Smith, and A. Oser.
Viability Of Listeria Monocytogenes On Commercially Prepared Hams Surfaced Treated With Acidic Calcium Sulfate And Lauric Arginate And Stored At 4°CSource: Meat Science, 71:92-99, September 2005, by Luchansky, J. B., J. E. Call, B. Hristova, L. Rumery, L. Yoder, and A. Oser
Components
- L-arginine is an essential amino acid present in many food sources, including poultry, seafood, cheese, cereals, nuts, seeds.
- Lauric acid can be found in oils such as coconut and palm kernel.
- Ethanol is the product of the fermentation of sugars. It is short-chain alcohol.
Lauric Arginate is a food-grade antimicrobial synthesized from food components lauric acid, naturally occurring in palm and coconuts, and L-arginine, an essential amino acid. Its remarkable antimicrobial properties, paired with low toxicity levels, make it an optimal bactericidal control tool in food production. This unique antimicrobial agent made from natural ingredients is proven to be extremely effective and can kill up to 99.99% of the foodborne pathogens and spoilage bacteria in a wide range of products.
Protect Your Products From Pathogenic Bacteria
Dealing with food recalls due to contamination results in profit losses and a public relations nightmare for any food brand that can lead to a stock market drop, losing the market share, and permanently damaging the brand in the consumers' eyes. You can protect your company from these misfortunes by adding a safe antimicrobial (LAE) as a bacterium kill step in your production process. Benefits of using LAE in your food production process:
- Focus on producing a high quality / wholesome product safe from pathogenic bacteria
- Regulatory Compliance
- Eliminate risks associated with product contamination (product loss, product recalls, and associated expenses)
- Secure Your Business and Brand Image

A&B Ingredients And CytoGUARD® LA Advantage
- A&B Ingredients is the premier manufacturer of Lauric Arginate in the U.S.
- We have extended expertise in the use and application of Lauric Arginate in meat and other food products
- We have collaborated with the USDA and many research institutions to substantiate the use of LAE as a food safety and food-protecting agent
- We have collaborated with the USDA to develop the SLIC® (Sprayed Lethality in Container) patented process for LAE application, which is exclusively licensed to A&B Ingredients, Inc.
- Regional and top 100 meat processors in the U.S. have adopted the SLIC® method
- Over ten years of experience with LAE supported by internal and third-party studies published in peer-reviewed publications
- Patented manufacturing process
- Product manufactured in the U.S.
- A diverse portfolio of food safety solutions that, when used in combination, can add a higher level of antimicrobial protection for your products
- Customized and proprietary solutions that improve efficacy and offer cost advantages
- Technical support and application testing
Related Resources
Get Expert Support and Formulation Guidance
Connect with our technical team for product samples, application support, and customized recommendations for your specific needs.
CONTACT US


